What is a nuclear moderator?

By Dr. Nick Touran, Ph.D., P.E., 2007-04-21, Updated 2023-11-30, Reading time: 4 minutes
The moderator is a material intentionally placed in a nuclear reactor core to slow neutrons down from their initial fast speeds without first getting absorbed non-productively by the fuel. Adding a moderator allows nuclear reactors to sustain a neutron chain reaction with minimal or no enrichment.
In common water-cooled nuclear reactors, the moderator is the same thing as the coolant: water. When fast neutrons strike the hydrogen atoms in H2O, they slow down a lot (like a billiard ball striking another). There are other good moderators like deuterium, graphite, beryllium, and more.
Why moderate?
When an incoming neutron causes the nucleus of an atom to split, other neutrons are released at very high speeds. If at least one of these neutrons can be made to fission another fuel atom, a sustained chain reaction is possible. However, fission isn’t the only reaction that neutrons can cause. Non-fission neutron absorption by Uranium-238 or similar will often capture neutrons and prevent them from causing a fission.
As it turns out, the likelihood of a neutron causing a fission rather than a capture is higher when the neutron is moving slower.
When neutrons collide with atoms, they slow down. Due to conservation of momentum, when a neutron strikes a heavy atom, it slows down a little (imagine a ping-pong ball bouncing off a bowling ball). When a neutron strikes a light atom of similar mass to the neutron itself, it slows down a lot (imagine a ping-pong ball bouncing off another ping-pong ball). Nuclear fuel atoms are heavy, so to get neutrons to slow down faster, you add light-weight atoms as moderators.
You could just mix your moderator and fuel together, but it’s dramatically better to keep them separate. If they’re closely mixed, neutrons would be absorbed by fuel as they are slowing down in neutron-capture reactions that compete with fission (the spiky part in Figure 1).
If you instead have a fast neutron first move into the separate moderator area, it will slow down “in peace,” i.e. away from the strong neutron absorbers. It will then re-enter the fuel as a slow neutron where the probability of fission is extremely high. The moderator effectively teleports neutrons from fast to slow and spits them out in the fuel right where they can best cause fission.
This ingenious “fuel lumping” insight was required in the first reactor, CP-1, which used natural uranium. There was no enriched uranium back in the early 1940s.
Choosing a moderator material
Some materials are better at slowing down neutrons than others. Conservation of energy and momentum laws explain that a neutron (mass 1) cannot slow down much after a collision with a heavy nucleus, like a fuel atom (mass 235). However, in a collision with Hydrogen (mass 1), a neutron can in fact slow down very far. This effect is often quantified by a parameter called \(\xi\), the mean lethargy gain per collision.
The number of collisions matters as well, which is proportional to the density of the moderator and its scattering probability. The moderating power is called \(\xi \Sigma_s\).
Moderators must also not be strong neutron absorbers. So we often divide by the neutron capture cross section to make an ideal figure-of-merit for moderators: \(\xi \Sigma_s/\Sigma_a\). These parameters are summarized for a number of moderators below:
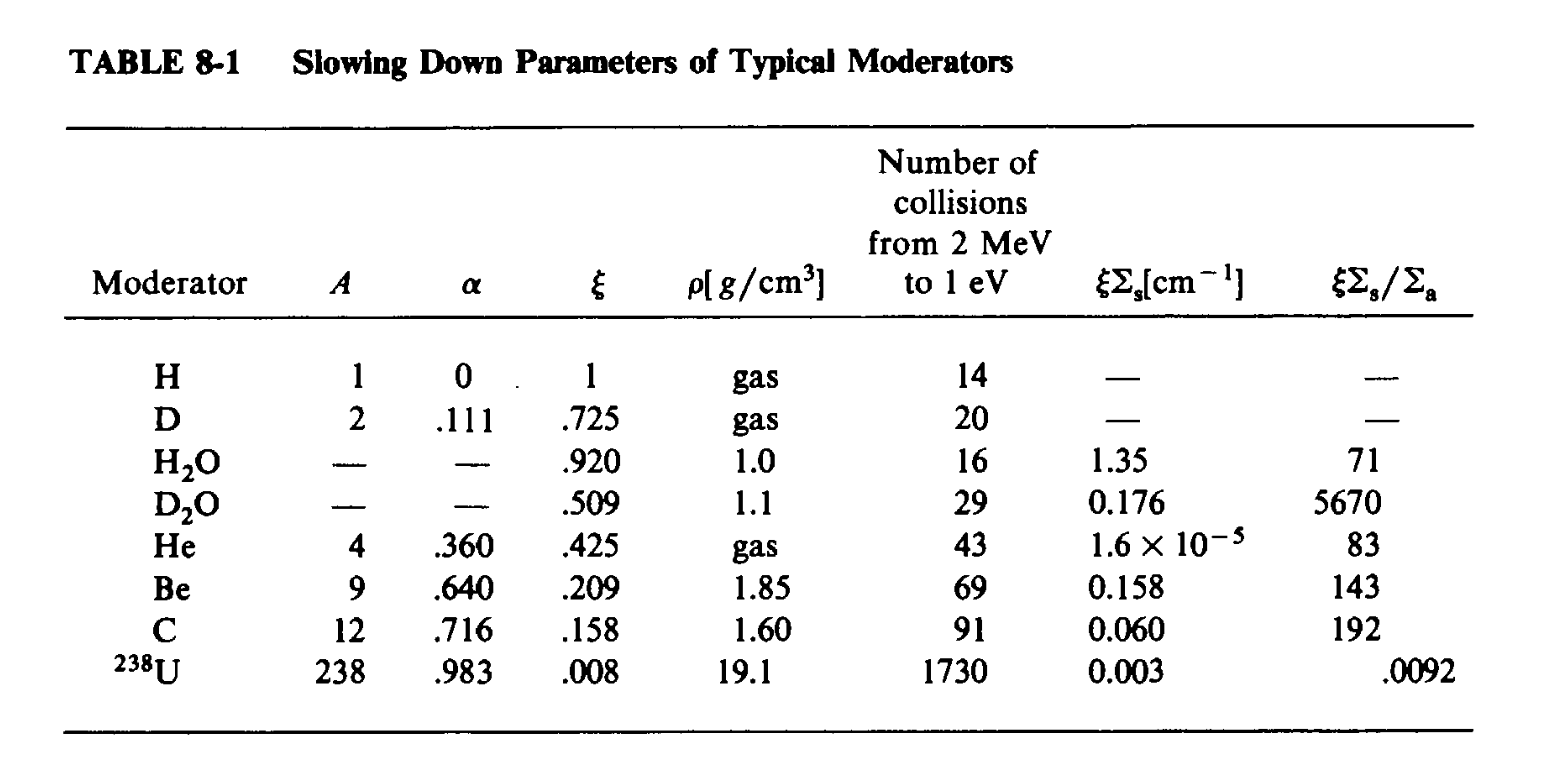
As you can see in the last column, deuterium (heavy hydrogen) is by far the best moderator. But regular hydrogen (aka light hydrogen) in water is OK. This is why deutrerium reactors (like CANDU) can run without enrichment but light-water reactors cannot.
Why are moderated reactors called thermal reactors?
The word thermal in thermal reactors indicates that the neutrons causing the most fissions are in thermal equilibrium with their surrounding atoms. All atoms vibrate based on their temperature, and so when neutrons slow all the way down, they don’t stop, but rather get jostled around by the atoms they are around. So thermal reactors is just a technical way of saying that the nuclear reactors has slow neutrons in it, slowed down by a moderator.
The alternate to thermal (aka slow-neutron) reactors are fast reactors, which intentionally do not have moderators placed in them. These reactors can get more total energy out of their fuel, but you have to use much higher enriched fuel to get them started and keep them going. That’s the price you pay for not having a moderator.
